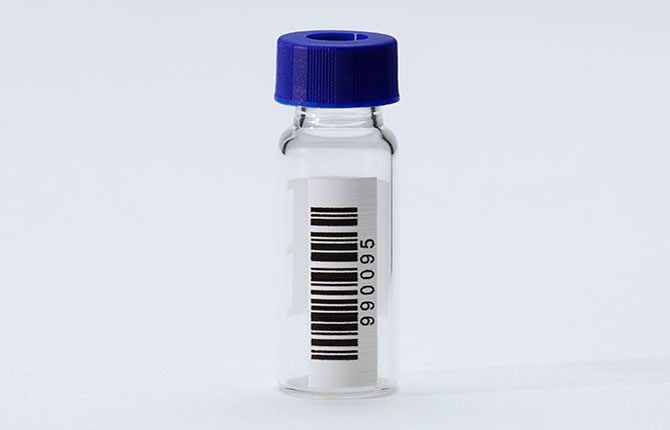
Sample management and workflow efficiency is critical in the laboratory. An accurate labeling system goes a long way toward facilitating a structured and efficient research environment.¹ Using sample tracking technologies such as 1D and 2D barcodes ensures sample integrity, reduces the loss of samples due to mislabeling, assists with storage and safety, and provides a way to track samples and sample-related information.
Automation continues to grow in research environments requiring the use of barcodes on sample containers and labware.² Reducing errors while increasing throughput, especially in terms of sample identification, is key as more automated processes are utilized.
Laboratory research and studies require collection, processing, analysis, and storage of many samples that are often handled by various departments. Workflows can be long with multiple steps and transfers in the process.
Labware preparation for research and study collection, processing and analysis is time consuming. Handwritten labeling or application of printed labels is challenging and prone to human errors (especially on small chromatography vials). To resolve these issues, we recommend removing label application from your lab, and instead ordering your chromatography vials with barcodes pre-applied.
In addition to eliminating a tedious step from your processes, pre-labeled containers can enhance your processes in other ways – keep reading for details on three ways your lab might benefit from making the switch to a pre-barcoded strategy.
Why use barcodes in chromatography?
The question of why barcodes should be used in chromatography is multifaceted, encompassing several critical aspects of laboratory operations. Compared to handwritten markings, a barcode system certainly has a lower margin for error – eliminating legibility factors and reducing opportunities for human error. For example, a big risk is placing a vial in the incorrect location in the autosampler or listing the sample in the incorrect place in the sequence table. Scanning provides better certainty as to what was placed where, significantly reducing that risk.
Furthermore, good laboratory practices point to proper sample and sub-sample tracking for each stage along the analytical process,³ as Ronald E. Majors points out. Dr. Majors also writes that “Proper identification of the collected primary sample by … application of a bar code for automatic reading … incorporation of RFI devices, or other means of documentation must be performed properly to ensure that later stages of processing can be traced unequivocally to the original primary sample.”
A proper sequence management system will further reduce the opportunity for error by seeking out duplicate and otherwise unusable barcodes post-printing. Systems like these can be a hassle to manage in-house, but any trustworthy supplier of pre-printed barcodes should have some system in place to ensure the quality of the barcodes you receive.
- Accuracy and Precision: Handwritten labels and notes are prone to errors, whether due to illegible handwriting, transcription mistakes, or human memory lapses. Barcodes offer a reliable and standardized method for sample identification, minimizing the risk of errors and ensuring accurate and precise analytical results.
- Efficiency and Productivity: Manual data entry and sample tracking consume valuable time and resources, leading to inefficiencies in laboratory workflows. By automating sample identification through barcode scanning, laboratories can streamline processes, reduce turnaround times, and increase overall productivity.
- Data Management and Traceability: Barcodes facilitate comprehensive data management by providing a unique identifier for each sample. This allows laboratories to track samples throughout the entire analytical process, from sample preparation to result reporting, ensuring traceability and accountability.
- Sample Tracking and Audit Trails: In today’s regulatory environment, maintaining detailed audit trails is essential for compliance with quality standards and regulations. Barcoded samples enable laboratories to create comprehensive audit trails, documenting each step of the analytical process and providing a transparent record of sample handling and analysis.
- Knowledge Sharing and Collaboration: Standardized barcode systems facilitate seamless data exchange and collaboration among research institutions, academic laboratories, and industry partners. By adopting a universal barcode format, laboratories can easily share analytical data, compare results, and collaborate on joint research projects, fostering transparency and reproducibility in scientific research.
How to use barcodes effectively in chromatography
Implementing barcode systems in chromatography requires careful planning and execution to ensure optimal performance and integration with existing laboratory workflows. Here are key steps to effectively use barcodes in chromatography:
- Barcode Labeling: Assign a unique barcode to each sample vial and associate essential information such as sample ID, batch number, and preparation date to the metadata in your LIMS. Use standardized barcode formats such as Code 128 or Data Matrix codes to ensure compatibility with barcode readers and software systems.
- Autosampler Integration: Select autosamplers with barcode readers or scanners capable of decoding barcode information from sample vials. Ensure seamless integration with autosampler software to enable automatic sample identification and tracking. There are several barcode-enabled autosamplers on the market, some of our favorites include: Gerstel-MPS, Shimadzu HS-20, CTC PAL RSI and RTC, and these models from Agilent.
- Training and Adoption: Provide comprehensive training to laboratory staff on the use of barcode technology in chromatography. Emphasize the importance of adherence to standardized procedures for barcode labeling, scanning, and data entry to ensure consistent and reliable results.
Where can I find pre-barcoded chromatography vials?
You can source pre-barcoded labware from certain labware manufacturers or from your identification and tracking service provider.
It’s fairly common for labware manufacturers to offer pre-marked labware in their catalog, however, options for chromatography applications are often minimal.
Today, the most reliable resource for pre-barcoded chromatography vials is working with a labware service provider to build a custom solution. Placing your vial labeling in the hands of labware labeling experts also offers additional perks, such as higher quality print options and precise automated application without investing in costly equipment yourself.
Our Labware Prep™ Services solution offers these additional services as well:
Labware Procurement
We understand that in the lab, it’s not only important to have the right label for the job, but the right labware. We maintain connections with major labware manufacturers in order to ensure our customers get the labware they need. Just let us know your preference and we’ll take care of placing the order.
Nase-Controlled Production
Rest assured, your labware is meticulously unpacked, marked, and re-packaged within our RNase/DNase-free environment, ensuring that your pre-labeled labware is ready for use when avoiding DNA or RNA contamination is crucial. This allows your staff to focus on their primary tasks.
Barcoding, Marking, Labeling
Our team of experts will use our in-house automation to apply adhesive barcode labels to your labware, or you may take advantage of our advanced marking technologies for a more permanent identification solution, depending on your needs and environment.
Tare-Weighing, Kitting
Upon request, our team can take on specific prep tasks, including tare-weighing, capping or de-capping, plugging, racking, and sorting tasks. Your labware and kits are assembled to your bespoke specifications, including brand standards, and you will receive your labware immediately, ready to use upon delivery.
Order fully prepared chromatography vials
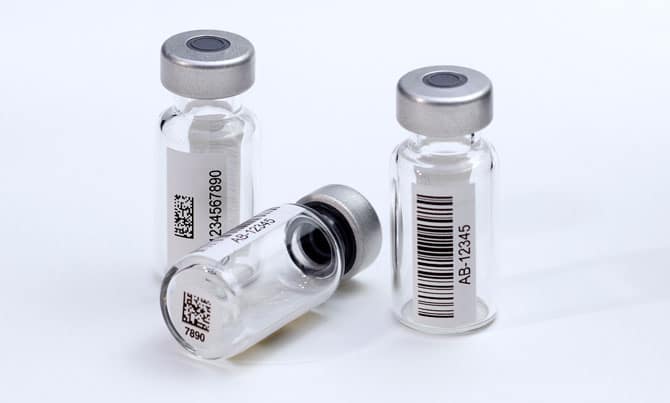
We offer two prepared chromatography kits to promote accuracy and efficiency in your laboratory. Each kit includes 100 pre-barcoded glass vials and their accompanying closures, conveniently packaged for easy use.
We utilize our cured ink direct mark barcode technology to ensure scannability and permanence.
These kits can significantly reduce the amount of time dedicated to prep work in your lab and protect your samples through HPLC, GC, and MS analysis. To learn more and request a sample, view our pre-barcoded chromatography kits here.
Final thoughts
Barcodes are being introduced to the chromatography process to improve the tracking and identification of samples. While printed barcodes are much simpler to prepare than handwritten markings, we want you to be aware that eliminating label application from your facility is an option that’s available to boost efficiencies even further.
In addition to reduced effort on your part, your partner will likely be able to ensure you receive barcodes with high enough resolution to be scanned consistently despite their small size. Improved durability and precision placement can further enhance your chromatography vials’ ability to withstand harsh exposures and interact with automated equipment.
If you’re considering barcodes for your 12×32 autosampler vials or even larger headspace vials, take a moment to evaluate whether outsourcing services offer benefits that align with your needs, challenges, and goals. If you have questions about our offerings, contact us, or give this blog post a quick read.
1. Janzen W, Admirand E, Andrews J, et al. (2019) Establishing and Maintaining a Robust Sample Management System. SLAS Technology. 24 (3), 256-268.
2. Liscouski, J. (2018). Some considerations in the automation of laboratory procedures. Unpublished. https://doi.org/10.13140/RG.2.2.27991.73125
3. Majors, R. (2013) Sample Preparation Fundamentals for Chromatography. Agilent Technologies Inc. 5991-3326EN